PMID: 26692646
This article has been cited by other articles in PMC.
Small incision cataract surgery (SICS) is one of the cataract surgical techniques commonly used in developing countries. This technique usually results in a.
Cataract is the most common cause of blindness. Cataract extraction with intraocular lens implantation is one of the most frequently performed and most effective surgical procedures in the field of medicine, worldwide.
Small-incision cataract surgery (SICS) is also called manual small-incision cataract surgery (MSICS) or sutureless extra-capsular cataract extraction (SECCE). It is a safe, cost-effective procedure with very good outcomes. The technique is well described in the literature, as is the management of its complications.
The quality of SICS, and most importantly the outcome for the patient, can be excellent. The World Health Organization (WHO) advises aiming for post-operative outcomes of at least 80% good presenting vision or at least 90% with best-corrected vision, and this is attainable with SICS. But with such a good procedure at your fingertips, how do you aim for and achieve quality? And how do you plan for, maintain, and monitor the best possible outcomes for your patients?
Training and learning
It is important to learn, to want to learn, and to maintain a good technique. There are many good resources available.
- MSICS classroom: http://classroom.globalsight.org
- Eye Surgery in Hot Climates 4th Edition (JP Publishers, 2015).
- Standard Operating Procedure Manual for Modern Small Incision Cataract Surgery (Tilganga Eye Centre): available via the Global Sight Alliance website: www.globalsight.org
- Aravind Eye Hospitals’ book Manual Small Incision Cataract Surgery: available as a free iBook download from iTunes: http://tinyurl.com/pu757w7
- Sutureless ECCE (2nd Edition) video: www.youtube.com/watch?v=LszyZqqR5v4
Time spent reading and watching videos again and again is time well spent. However, a good teacher is critical. It is important to be able to practise and get feedback. There is a big role for simulation in training and practice – either ‘wet-lab’ as in the use of animal eyes to learn and practise; or ‘dry-lab’ as in the use of artificial eyes and simulators (HelpMeSee.org and simulatedocularsurgery.com).
Evaluation and selection for surgery
As a surgeon, you may not see all of your cataract patients before surgery. If this is the case, it is imperative that the nurses working with you are diligent in their pre-operative assessment. All patients should have the following assessments: visual acuity, intra-ocular pressure (IOP), pupils (looking for a relative afferent pupillary defect, or RAPD), and past ophthalmic history. Selection for surgery is very important. Listing a patient for routine cataract surgery who has an RAPD, high IOP, ora history of severe retinal disease may lead to a poor outcome. The patient should be fully assessed, and appropriate consent taken before a ‘guarded prognosis’ is made. In addition, for good vision outcomes, it is essential to have accurate biometry and to have a large range of intraocular lenses available.
Surgical technique
The procedure itself is wonderful when all goes well; however, every eye is different, every operation is different, and each and every step of the procedure is as important as the one before.
A good draping is necessary to capture the eyelashes, especially those of the upper lid.
The superior rectus suture is important: it immobilises the eye and assists with scleral tunnel ‘opening’ when extracting the nucleus. Going too deep with the needle may penetrate the eye, and going too shallow will engage the conjunctiva only.
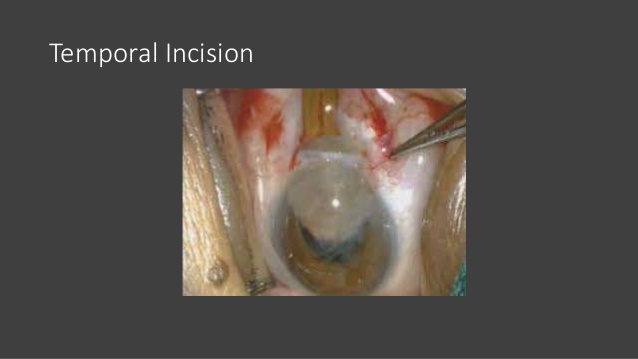
Firm scleral fixation (throughout the tunnel construction) should be maintained by using good forceps (Figure (Figure11).
The scleral tunnel is very important. A very curved and ‘frown-shaped’ incision should be made initially. If the incision is too flat, this will induce a significant against-the-rule astigmatism. Use adequately sharp blades.
When forming the tunnel with the crescent blade, aim to see just enough of the metal of the blade. If you cannot see any of it, you are too deep and will likely prematurely enter the anterior chamber. If you see too much, a buttonhole will form. Dealing with complications in tunnel construction may be necessary: a button hole may lead to leakage and should be undermined in a different plane or a new entry site fashioned. Premature entry into the anterior chamber will often require a suture.
The capsulotomy can be linear or continuous-curvilinear. Lots of small puncture marks are necessary for a linear capsulotomy. Aim for just above the halfway line. This will leave a good inferior portion to protect the corneal endothelium when extracting the nucleus, but also enough of a superior portion to support a sulcus-placed IOL if the posterior capsule is ruptured and cannot support an IOL.
Thorough hydro-dissection helps mobilise the nucleus. Always check that the cannula is on tightly before entering the eye. Lift the capsule slightly when injecting underneath it.
The most difficult part of the procedure is the mobilisation of the nucleus. Once you are happy that the nucleus is free in the bag, inject visco-elastic into the anterior chamber to protect the endothelium. Use the cannula, while slowly injecting visco-elastic, to dislodge the upper equator of the lens nucleus. The important point is to press backwards and slightly down within the scleral wound beyond the upper equator, such that the upper part of the nucleus actually starts to move forward rather than backwards.
Inject a good amount of visco-elastic behind the nucleus to push back the posterior capsule before inserting the vectis or fishhook needle to extract the nucleus.
Once the nucleus is removed, take great care when removing the soft cortical lens matter. Increase the magnification on the microscope for this stage, as well as for the capsulorhexis.
An injection of antibiotic into the anterior chamber (intra-cameral) should be performed at the end of the procedure (with either cefuroxime (1 mg in 0.1 ml) or moxifloxacin, but only if you can guarantee that the concentration will be correct every time. This may help to prevent postoperative endophthalmitis, but can severely damage the corneal endothelium if an incorrect dosage is injected.
Complications and their management
Dealing with vitreous loss is important. It is most important that no vitreous remains in the anterior chamber going up to a wound or paracentesis. This is certain to invite future infection, chronic macular oedema, or even a retinal detachment. It should almost always be possible to place either a posterior chamber or sulcus IOL. Avoid an anterior chamber IOL if at all possible.
If there is any doubt about the wound, or if the initial incision is too flat, then place a 10-0 nylon suture in the main incision.
Post-operative treatment and evaluation
Patients must be given a combination of antibiotic and steroid drops for a few weeks after surgery. It is important to ensure that patients understand the importance of using the drops, and also how to use them.
Figure 1.

Stabilising the sclera with toothed forceps while making a frown-shaped incision
Post-operative evaluation is critical. Examine patients on the first postoperative day to diagnose and treat surgical complications. Vision assessment and ideally refraction should take place at 4–6 weeks after surgery and are essential for assessing surgical outcomes. Patients and/or carers must be educated about possible complications. They must be told what to do and where to go if they have untoward symptoms. This will help to ensure early diagnosis and treatment of complications such as intraocular infection.
Audit
The value of being interested in, recording, monitoring and then reflecting on all your surgical outcomes cannot be over-emphasised. On a very personal level, as a surgeon, this will improve your selection, technique and management; ultimately, it will make you a better surgeon and improve outcomes for the patients that you treat. Audit of outcomes should be absolutely mandatory everywhere. It may seem like an intrusion or extra effort, but it definitely makes you a better surgeon.
Conclusions
Ultimately, the best surgical safety, technique, and outcomes for your patient are not up to a textbook, DVD, website or trainer. They are in your, the surgeon's, hands. Only you can strive for the best surgical outcome, and the eye team can help you to achieve this.
Further reading
1. Gurung R, Hennig A.Small-incision cataract surgery: tips for avoiding surgical complications.Comm Eye Health J 288; 21(65): 4–5. [PMC free article] [PubMed] [Google Scholar]
2. Sandford-Smith J.Sutureless cataract surgery: principles and steps.Comm Eye Health2003; 16(48): 49–53. [PMC free article] [PubMed] [Google Scholar]
3. Hennig A.Sutureless non-phaco cataract surgery: a solution to reduce worldwide cataract blindness?Comm Eye Health J2003; 16(48): 49–51. [PMC free article] [PubMed] [Google Scholar]
4. Yorston D.Using intracameral cefuroxime as a prophylaxis for endophthalmitis.Comm Eye Health J2008; 21(65): 11. [PMC free article] [PubMed] [Google Scholar]
Articles from Community Eye Health are provided here courtesy of International Centre for Eye Health
PMID: 18504466
This article has been cited by other articles in PMC.
Small incision cataract surgery (SICS) is one of the cataract surgical techniques commonly used in developing countries. This technique usually results in a good visual outcome and is useful for high-volume cataract surgery.,,
This article describes how to minimise surgical complications in SICS.
Before you begin
With SICS, as with all cataract surgery techniques, it is mandatory to perform a thorough preoperative assessment of the patient (see article on page 12). This will allow the surgeon to prepare for anticipated complications – for example, a dislocated or subluxated lens – and to plan the operation accordingly.
Prepare the patient in the following way:
- Wash the patient's face.
- Instil povidone-iodine (Betadine) 5% aqueous eye drops (Figure (Figure11).Instilling povidone-iodine (Betadine) 5% eye drops
- Clean the skin around the eye with povidone-iodine 10% (Figure (Figure22).Skin around the eye cleaned with povidone-iodine (Betadine) 10%
Other measures will also help to reduce the risk of postoperative endophthalmitis: proper hand washing (see ‘how to’ article on page 17), the use of sterile instruments, the ‘non-touch’ technique, the subconjunctival injection of antibiotics,, and the intracameral injection of cefuroxime at the end of surgery (see article on page 11). The dose of intracameral cefuroxime must be meticulously prepared, as no commercially made preparation is available (see box on page 11).
Tunnel construction
Tunnel size
Tunnel size The expected size and density of the nucleus should determine the size of the tunnel. For example, the extraction of immature cataracts in younger patients may only require a small tunnel, just large enough for the intraocular lens (IOL) optic to pass through. Very big, brown nuclei require a larger tunnel size. These nuclei can sometimes be up to 8 mm in diameter and 4 mm thick. However, a large tunnel need not be a problem: even larger tunnels are self-sealing and don't need suturing if they are prepared correctly. If there is doubt about the self-sealing effect, the surgeon may apply one or two sutures at the end of surgery. If correctly tied, these will, at the same time, reduce any induced astigmatism.
Constructing the tunnel
- Only a correct sclerocorneal tunnel incision, at least 1 to 2 mm into the clear cornea, leads to a self-sealing wound.
- Scleral cauterisation before tunnel. construction reduces the risk of pre- and postoperative hyphaema.
- Sharp tunnel instruments (such as the crescent knife and keratome) should be used to construct the tunnel. A blunt keratome could cause stripping of Descemet's membrane.
- Stabilising the sclera with toothed forceps makes tunnel construction easier (Figure (Figure3).3). However, in order to avoid tunnel damage and leakage, the forceps should not be used on the tunnel flap.Stabilising the sclera with toothed forceps
- With a half-thickness sclerocorneal tunnel incision, the direction of the crescent knife should always be parallel to the sclerocorneal plane.
- Judge the depth of half-thickness sclerocorneal tunnel incisions by observing how clearly you can see the crescent knife during the incision (Figure (Figure4).4). If the crescent knife can be seen very clearly, this indicates that the scleral layer is very thin and that the crescent knife might perforate to the outside. (causing what is known as a ‘buttonhole’)Making the sclerocorneal tunnel incision
- A buttonhole can be corrected by making a deeper ‘frown’ incision and dissecting the tunnel in a deeper plane, starting at the opposite side of the buttonhole.
- If the crescent knife is not visible during the incision, this indicates that you are working too deeply inside the sclera; you may perforate towards the anterior chamber's angle (a ‘premature entry’).
- A premature entry could lead to surgical complications, such as iris trauma or iridodialysis, iris prolapse, and a tunnel which is not self-sealing.
- Manage a premature entry by starting a more shallow dissection at the other end of the tunnel. Suturing of the wound is required at the end of surgery.
Opening of the anterior capsule
This can be done by different techniques (such as linear capsulotomy (Figure (Figure5),5), the ‘can-opener’ technique, and triangular or V-shaped capsulotomy) or by capsulorhexis.
Linear capsulotomy
Capsulotomies are easy to perform, but may lead to uncontrolled capsular tear extension, posterior capsule rupture, vitreous loss, and IOL decentration. These problems can be avoided by a careful hydrodissection, especially in patients with posterior polar cataract or posterior lenticonus (hydrodissection is most effective if the fluid is injected directly into the capsule). Keeping instrument manipulation to a minimum during surgery will also help you to avoid posterior capsule rupture.
The best capsular opening is a continuous curvilinear capsulorhexis (CCC): it will guarantee a long-term, ‘in the bag’ IOL centration (Figure (Figure6).6). However, CCC is more difficult to learn. This technique sometimes requires staining of the capsule and the opening also needs to be large enough for the nucleus to get through. It may therefore not be possible to use this technique in eyes with very big nuclei and smaller pupils. In such a situation, a linear, triangular, or other capsulotomy may be preferred.
Continuous curvilinear capsulorhexis
Failing to complete the anterior capsulotomy, making a too-small CCC, and pulling residual anterior capsular tags can cause the posterior capsule to rupture. Early recognition and correction of these problems is very important to avoid further complications.
Nucleus removal
During SICS, different techniques can be used to remove the nucleus: either hydroexpression alone (using an anterior chamber maintainer), hydroexpression plus extraction (using an irrigating vectis or Simcoe cannula), or extraction alone (using a ‘fishhook’ needle). Problems with these different SICS techniques are mainly related to the size of the tunnel and the proximity of the nucleus to the corneal endothelium.
Difficulties with nucleus delivery are mostly due to the inner tunnel opening being too small. This should be checked before nucleus removal, e.g. with the visco cannula (Figure (Figure7).7). If there is any doubt about the correct tunnel size, it is better to further enlarge the tunnel before removing the nucleus. However, the surgeon should avoid cutting into the anterior chamber's angle while enlarging the inner tunnel opening, as this carries an increased risk of hyphaema.
Checking the tunnel size
While lifting the nucleus into the anterior chamber (Figure (Figure8),8), special care is required in patients with pseudoexfoliation and in older patients with weak zonules.
Mobilising the nucleus before lifting it into the anterior chamber
While delivering the nucleus through the tunnel, accidental contact between the nucleus and the corneal endothelium must be avoided. Otherwise, postoperative corneal oedema, and sometimes even corneal decompensation, may occur.
In order to avoid such corneal problems, you must inject sufficient viscoelastic fluid between the lens and the cornea to protect the endothelium. Instruments for nucleus removal, such as the irrigating vectis, Simcoe cannula, or fishhook, should be kept away from the cornea and should not push the nucleus against the cornea during nucleus delivery.These instruments should push slightly posteriorly, which will help to open the incision for easier nucleus delivery (Figures (Figures99 & 10). In addition, gently pulling the bridle suture makes nucleus delivery through the tunnel easier.
Inserting the fishhook needle
Delivering the nucleus
Removal of the cortex
Most of the lens cortex can be removed with a Simcoe cannula through the tunnel (Figure (Figure11).11). A sub-incisional cortex can be safely aspirated through a side port at 130–180˚ from the incision site. If stripping of Descemet's membrane occurs while cleaning the cortex, great care should be taken not to tear it off. If this happens, air should be injected into the chamber at the end of the operation to push Descemet's membrane against the cornea.
Removing the cortex using a Simcoe cannula
While clearing the cortex with a Simcoe cannula, posterior capsule rupture and vitreous loss may occur. This can be avoided by carefully watching the posterior capsule. Wrinkles indicate that the posterior capsule is caught in the aspiration port of the Simcoe cannula. This requires immediate backflushing to avoid posterior capsular rupture.
To reduce the risk of a postoperative increase in intraocular pressure, thorough removal of viscoelastics is required.
Contributor Information
Reeta Gurung, Deputy Medical Director, Tilganga Eye Centre, Kathmandu, Nepal. Email: moc.liamg@gnurugateer.
Albrecht Hennig, Programme Director, Eastern Regional Eye Care Programme, Lahan, Nepal. Email: ten.xmg@ginnehka.
References
1. Hennig A, Kumar J, Yorston D, Foster A. Sutureless cataract surgery with nucleus extraction: outcome of a prospective study in Nepal. Br J Ophthalmol. 2003;87:266–270.[PMC free article] [PubMed] [Google Scholar]
2. Hennig A, et al. World Sight Day and cataract blindness. Br J Ophthalmol. 2002;86:830–31.[PMC free article] [PubMed] [Google Scholar]
3. Ruit SPG, Gurung R, Tabin G, Moran D, Brian G. An innovation in developing world cataract surgery. Clin Exp Ophthalmol. 2000;28:274–279. [PubMed] [Google Scholar]
4. Ng JQ, Morlet N, Bulsara MK, Semmens JB. Reducing the risk for endophthalmitis after cataract surgery: population-based nested case-control study: endophthalmitis population study of western Australia sixth report. J Cataract Refract Surg. 2007;33(2):269–80. [PubMed] [Google Scholar]
5. Kamalrajah S, Ling R, Silvestri G, Sharma NK, Cole MD, Cran G, Best RM. Presumed infectious endophthalmitis following cataract surgery in the UK: a case-control study of risk factors. Eye. 2007;21(5):580–6. [PubMed] [Google Scholar]
6. ESCRS Endophthalmitis Study Group. Prophylaxis of postoperative endophthalmitis following cataract surgery: results of the ESCRS multicenter study and identification of risk factors. J Cataract Refract Surg. 2007;33(6):978–88. [PubMed] [Google Scholar]
7. Schroeder B. Sutureless cataract extraction: complications and management; learning curves. Comm Eye Health J. 2003;16(48):58–60.[PMC free article] [PubMed] [Google Scholar]
8. Traianidis P, Sakkias G, Avramides S. Prevention and management of posterior capsule rupture. Eur J Ophthalmol. 1996;6(4):379–82. [PubMed] [Google Scholar]
Articles from Community Eye Health are provided here courtesy of International Centre for Eye Health
